|
11/8/2023 0 Comments Malate oxaloacetate shuttle
In animals, different types of cells die in many different ways. 1 One plausible explanation is that cell death caused by each mutant is propagated in different cell types, with distinct underlying mechanisms. is that cell death induced by mod1 has a mosaic distribution in the leaf, whereas the lesion mimic mutants, such as lsd1, cause a runaway cell death. 1 Another interesting observation by Zhao et al. 1 It is puzzling that proteins required for mod1-triggered cell death are not required for the cell death induced in lesion mimic mutant plants. 
2 Lesion mimic mutants, such as lsd1, can induce autoimmunity and HR in plants, which is enhanced by oxidative stress. One well-known biotic stress-induced cell death is the hypersensitive response (HR), which is usually associated with activation of plant immunity. 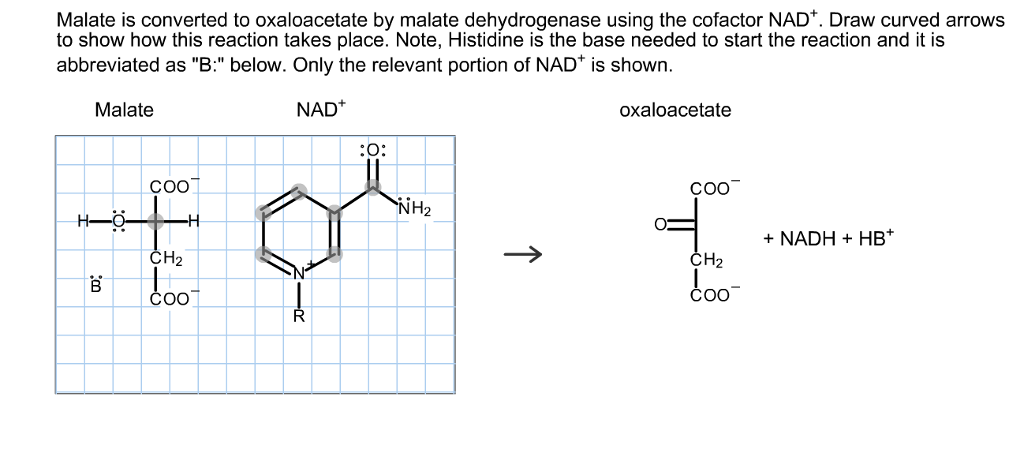
Cell death in plants can be either developmentally induced or triggered by environmental stresses, such as biotic and abiotic challenges. Plants have evolved mechanisms to control cell death and avoid collateral damage. 1Ĭell death occurs throughout plant life. These processes couple with the conversion between NADH (NAD hydrogen) and NAD + (an oxidizing agent) and allow organelles in plants to communicate through the shuttling of malate to control ROS levels and redox status. In mod1 mutant, pINAD-MDH reduces OAA to malate in chloroplasts, and mMDH1 oxidizes malate to OAA in mitochondria. DiT1 resides in the chloroplast envelope and one of its putative functions is to export malate from chloroplasts to the cytosol, whereas mMDH1 localizes to mitochondria. 1 The authors then verified that plNAD-MDH protein is localized in chloroplasts, the same as MOD1. also confirmed that the impairment of fatty acid synthesis in mod1 mutant can induce malate accumulation in chloroplasts, which subsequently causes oxidative stress in both chloroplasts and mitochondria. These proteins are all key components of the malate/oxaloacetate (OAA) shuttle in plants. 1 They reported new mutations in genes that encode a plastidial nicotinamide adenine dinucleotide (NAD)-dependent malate dehydrogenase (plNAD-MDH), a chloroplastic dicarboxylate transporter 1 (DiT1) and a mitochondrial malate dehydrogenase 1 (mMDH1). screened for additional mod1 suppressors with intact mETC complex I activities. So the molecule is not split in two, because a bond remains linking the two atoms that got either the H+ or OH- added to them.To test this hypothesis, Zhao et al. In this way an OH- has been added to one part of the original molecule, and a H+ to the other, and the molecule has been split (lysed) into two by water(i.e hydrolysed).īut in step 7, fumarate has a double bond across which the water is added (in a 'hydration' reaction). It then picks up the spare H+ from the original water. In hydrolysis reactions, the negatively charged oxygen of water 'attacks' some partially positive charged atom on another molecule and shares its electrons with it in a new bond, and a 'leaving group', which is happier to take its electrons with it, is displaced and leaves. That being said, from a chemical point of view, fumarate and water reacting the way they do makes sense, whereas I don't see how a hydrolysis reaction could take place. So enzymes are often responsible for deciding at exactly which position in a large molecule a reaction will happen, where in a test tube, the chemicals might slightly prefer to react at a different position. One thing that determines which reactions happen in a cell is which enzymes there are, since they are the catalysts that increase the rate of feasible reactions so that they actually happen at a useful rate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
